Details
Why Assess Homocysteine Levels?
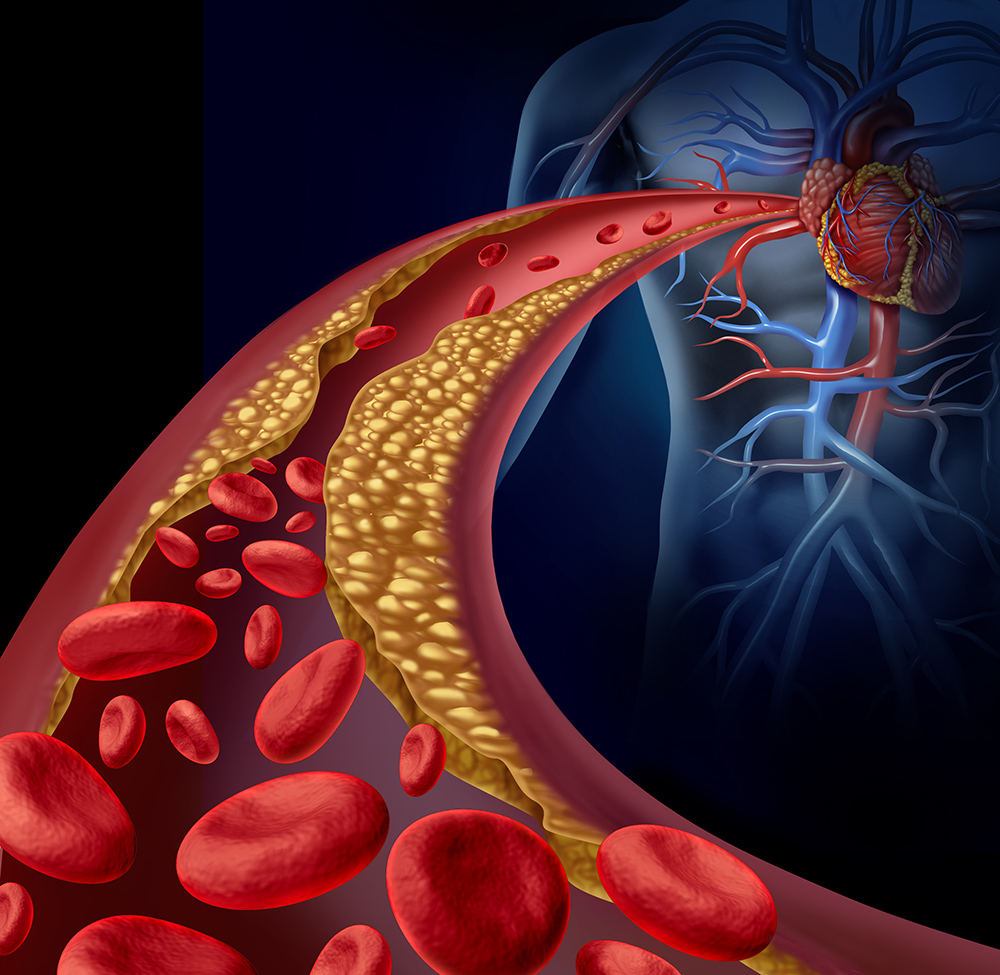
Homocysteine is an amino acid containing sulfur that is not used for protein synthesis. It is formed during the methionine cycle, also known as the transsulfuration methylation cycle, which plays a crucial role in linking sulfur amino acid metabolism with important methylation reactions. Homocysteine can be produced by breaking down S-adenosylmethionine or by converting homocysteine thiolactone, a toxic substance, to homocysteine using paraoxonase 1 (PON1), an enzyme that also helps detoxify organophosphate pesticides.
To clear homocysteine from the body, methyltetrahydrofolate, methylcobalamin, betaine, or pyridoxal-5-phosphate is needed. Methylcobalamin is a highly effective methylating coenzyme that can directly convert homocysteine to methionine without the need for another enzyme.
Various factors can increase homocysteine levels, including genetic deficiencies in enzymes involved in the transsulfuration methylation cycle (such as MTHFR, CBS, BHMT, and others), a diet high in methionine, or deficiencies in folate, vitamin B12, vitamin B6, or betaine. Megadoses of niacin, nitrous oxide anesthesia, and excessive copper can also lead to elevated homocysteine levels.
On the other hand, a diet rich in folate compounds, vitamin B12, methylcobalamin, betaine, or supplements containing these nutrients can help lower homocysteine levels. Exposure to certain toxic chemicals can activate CBS, leading to increased conversion of homocysteine to cystathionine and a decrease in homocysteine levels. Removing toxic substances from the body may also result in increased serum homocysteine levels.
What are Testing Considerations?
An excess of cystathionine beta-synthase can significantly affect homocysteine levels and make it an unreliable marker of the transsulfuration methylation cycle. This is commonly seen in individuals with Down’s syndrome, where the CBS enzyme is overproduced due to the presence of an additional copy of chromosome 21. This leads to a reduction in homocysteine levels as the excess CBS channels homocysteine into the CBS reaction, limiting methionine production. In such cases, it is crucial to increase coenzymes like methyltetrahydrofolate, methylcobalamin, and the methyl donor betaine.
Exposure to toxic chemicals can also lead to an increase in CBS production, allowing the body to produce more glutathione to detoxify the toxins. This can result in low homocysteine values, rather than high values. Low homocysteine levels in autism may be associated with severe toxic chemical exposure, confirmed in many cases of autism. In such cases, the baseline homocysteine may be below 7.0, and serum homocysteine may increase after detoxification.
Organic acids testing can be helpful in such cases. The markers for excessive CBS activity (2-hydroxybutyric acid), toxicity (succinic acid), and glutathione deficiency (elevated pyroglutamic acid) can be used to determine the transsulfuration methylation pathway. 2-Hydroxybutyric acid is a downstream product of CBS activity and is usually high in Down’s syndrome and toxic exposure cases, while plasma homocysteine is low. Succinic acid is often high due to toxic inhibitory effects on succinic acid dehydrogenase in the Krebs cycle, and the glutathione deficiency marker pyroglutamic acid will be high in cases of toxicity.
Learn More About the Homocysteine Test: Explore FAQs
Analytes
This test will allow you to personalize patient care through determination of homocysteine levels.
Sample Reports
The Homocysteine Test report can help to determine if intervention is needed to prevent the toxic effects of excess homocysteine.
Test Prep and Instructions
MosaicDX offers patient-friendly sample collection kits that simplify testing. Our kits include visual, step-by-step instructions for test preparation and sample collection, personalized shipping cards, and pediatric collection bags if needed. With MosaicDX, patients can easily collect samples for testing with confidence and accuracy.